Saphenion science - vein glue allergy? is there a significant risk?
Received: 08-Jun-2020 Accepted Date: Jun 22, 2020; Published: 27-Nov-2020
This open-access article is distributed under the terms of the Creative Commons Attribution Non-Commercial License (CC BY-NC) (http://creativecommons.org/licenses/by-nc/4.0/), which permits reuse, distribution and reproduction of the article, provided that the original work is properly cited and the reuse is restricted to noncommercial purposes. For commercial reuse, contact reprints@pulsus.com
Abstract
In a few specialist comments, presentations and articles by phlebologically active colleagues is repeatedly warned against the emergence of an allergy to the cyanoacrylate used in the vein glue. Actually a presentation at UIPmeeting in Krakow (Poland).Keywords
Cyanoaccrylate allergy, Vein glue allergy, Allergy of VenaSeal®, Allergic symptoms vein glue.
Introduction
No allergic reaction in 94 months using vein glue
Our nearly 8 years experiences in using varicose vein glue did not show any allergies in all 2710 sealed truncal varicose veins. By default, we perform regular clinical and ultrasound follow – ups of treated veins. Until now we have not been reported allergic, nor have we found any clinical signs of allergies or allergic symptoms [1,2].
This affected all treated patients, including those who reported in the preoperative preparation and the discussions about allergies or as a multiallergy sufferer had a corresponding allergy pass [3,4].
As previously started, all our self – payer patients receive a warranty card on the effectiveness of the sealing vein procedure. This is a 3 – year warranty on the therapeutic effect and possibly free repetition of the glue associated. In this context of warranty, we have treated a second intervention with sealing glue in 24 patients [4,5].
This fact speaks against the conjecture of colleague Dr. Thun, that allergies may occur during recurrent interventions (vasomed 1/2019, pages 28 – 31). In the same sheet reported Dr. Neff of 56 glue – treated truncal varicose veins, also all without an allergy [6].
Vein Glue Allergy
Facts from scientific literature
We find in the last 35 years only 25 (!) papers on this subject. Not all articles describe allergic reactions. The majority of the articles were published by dermatologists and aesthetic medicine. All publications are case descriptions. Compared to the number of patients using a cyanoacrylate glue, these numbers are negligible [7-10].
This fact is very interesting because of - actually - over 140 000 patients treated with vein glue.
Essentially it is about the suspicion of allergic skin reactions / inflammation / foreign body – reactions in the closure of surgical wounds of skin medicine (tumors, fat tumors etc). However, the described cases are – in relation to the number of treated patients – purely case – by – case descriptions [11,12].
Sage Journal only contains individual case descriptions of foreign body reactions after VenaSeal® therapy. Systemic allergic reactions are not found here.
In PubMed was found an article with a suspected systemic allergy to VenaSeal® vein glue: https://www.ncbi.nlm.nih.gov/pubmed/31440717.
Furthermore, allergic contact dermatitis after therapy with the cyanoaccrylate vein glue was described in two cases.
Other reports come from the field of aesthetic medicine. For example, contact allergies have been described in the bonding of artificial fingernails and eyebrows. In some cases, allergic rhinitis and asthma attacks have been described. The latter were mainly noticed by the inhalation of adhesive vapors and skin contact [13].
Even when working with cyanoacrylate glue in model making, it has come in one case described an asthma attack.
Case Report
Vein Glue Allergy
In February 2020 we diagnosed an 84-year-old patient with a 4 ° CVI with a leg ulcer on the left lower leg. There were 2 defective varicose veins on both legs. Both the GSV and the SSV were considerably refluxive and expanded. There was marked tissue edema on both lower legs, the skin showed traces of chronic inflammation and recurrent soft tissue inflammation [15].
We recommended minimally invasive, nonthermal, nontumescent catheter therapy using the vein glue VenaSeal® - with the possibility of simultaneous therapy of all 4 insufficient truncal varicose veins [16].
Within a week the patient decided on therapy and on February 12th. we also carried out this simultaneously on the 4 truncal varicose veins.
The follow-up examinations by means of clinical examination and duplex ultrasound showed a clear decrease in soft tissue swelling and venous pressure in the lateral veins. The existing ulcer on the left lower leg was already smaller and showed a clean wound area with a clear tendency to heal. The ultrasound examination confirmed that all 4 varicose veins were almost completely closed [17,18].
Micro foam therapy for side branches varicose veins was then started 14 days later. We first treated the left leg to further accelerate the healing of the ulcer.
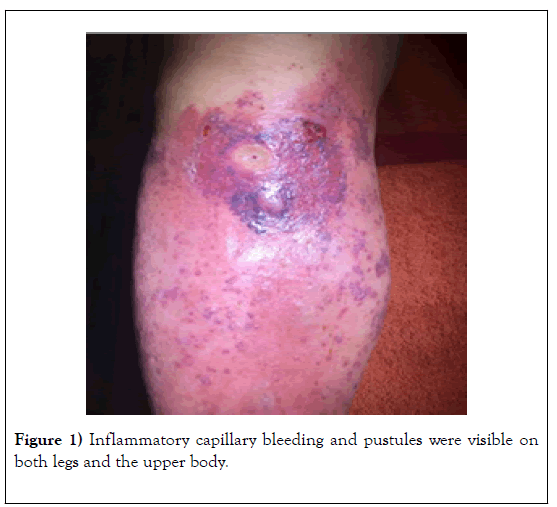
Another 14 days later the patient turned acute with a massive inflammatory reaction on the right lower leg in front. Inflammatory capillary bleeding and pustules were visible on both legs and the upper body (Figure 1).
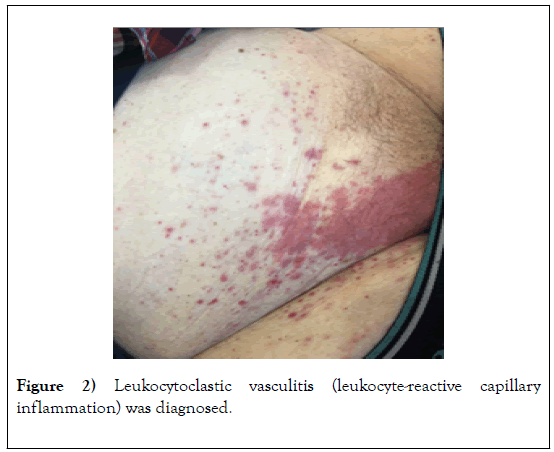
The patient was admitted to a university clinic on suspicion of a cyanoacrylate allergy after VenaSeal® - therapy. The inpatient stay lasted 6 days. Leukocytoclastic vasculitis (leukocyte-reactive capillary inflammation) was diagnosed (Figure 2). Tissue removal then showed suspicion of leukocytoclastic vasculitis of parainfectious genesis due to erysipelas on the right lower leg [19].
The therapy consisted of cooling, anti-inflammatory bandages on the vasculitis areas, and lavanide gel as well as fatty gauze for the ulcer. Antibiotic therapy was also carried out.
The duplex sonographic diagnosis of the leg veins showed no further evidence of persistent chronic venous insufficiency [20].
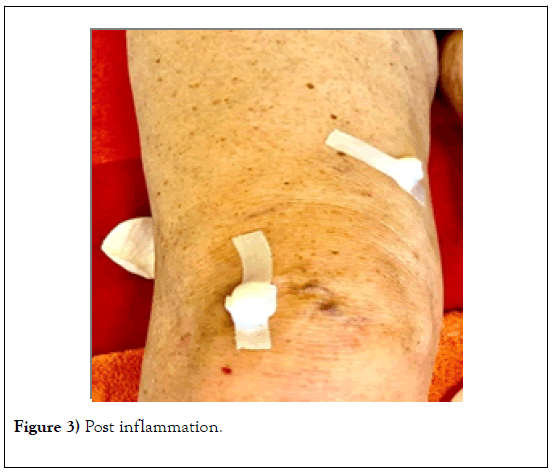
The treated varicose veins were effectively closed (Figure 3).
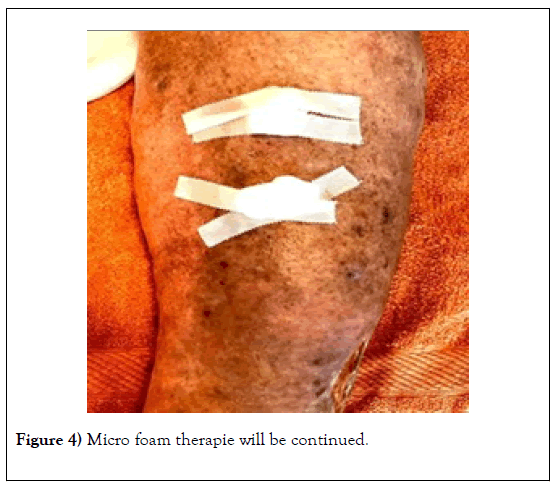
In the mean time we have micro foam therapy on the right leg continued, the ulcer on the left lower leg has become significantly smaller. A new ulcer has now formed on the right lower leg in the area of erysipelas (Figure 4).
Evidence of an allergy to the vein glue VenaSeal® could not be provided.
Discussion
Allergy potential of the vein glue overestimated
We believe that statements about the allergenic risk of vein glue are sometimes overstated. On the one hand, only very small amounts of the glue – approx. 1.2 - 1.8 ml per truncal varicose vein (in maximum 8ml in our Saphenion® Vein Care Centers) – are introduced into the vein via catheter. On the other hand, there are only very few individual case descriptions for the general topic „cyanoacrylate glue and allergy”.
In addition, hypersensitivity reactions (foreign body reaction) are possible. These appear as a red stripe across a vein segment treated with VenaSeal®. In our 2710 treated truncal varicose veins, these reactions occurred in 203 veins, which corresponds to a share of 7.5%.
The therapy for this side effect appears very simple. A cooling alcohol bandage twice a day over 3-7 days is completely sufficient. In no case was a medication or compression necessary for us. However, we start cooling immediately after treatment. On the operating table, we put on all our patients an alcohol bandage for about 30 minutes [21-22].
Since this very simple therapy we have decreased the hypersensitive foreign body reaction from 19% in the first 2 years using vein glue to 7,3% until today!
And we believe that this phenomenon occurs primarily in patients whose truncal veins are extra-anatomically outside the fascia, directly in the hypodermis. In this case, we point out this possible side effect particularly intensively [23].
Of course, as with any drug, an explanation of the problem of a possible allergy must be made. However, the risk of actually developing allergic reactions is negligible in sealing veins [24-27].
Conclusion
This must be communicated in any case. A blanket suspicion of allergic side effects is completely out of place.
Disclosure
The Author do not have any potential conflict of interests.
REFERENCES
- Navarro-Triviño FJ, Cuenca-Manteca J, Ruiz-Villaverde R. Allergic contact dermatitis with systemic symptoms caused by VenaSeal. Contact Dermatitis. 2020;82(3):185-87.
- Tang YT, Rathnaweera HP, Kam JW, et al. Endovenous cyanoacrylate glue to treat varicose veins and chronic venous insufficiency—Experience gained from our first 100+ truncal venous ablations in a multi-ethnic Asian population using the Medtronic VenaSeal™ Closure System. Phlebology. 2019;34(8):543-51.
- Jones AD, Boyle EM, Woltjer R, et al. Persistent type IV hypersensitivity after cyanoacrylate closure of the great saphenous vein. J VascSurg Cases Innov Tech. 2019;5(3):372-74.
- Gibson K, MinjarezR,Rinehardt E, et al. Frequency and Severity of Hypersensitivity Reactions in Patients After VenaSeal™ Cyanoacrylate Treatment of Superficial Venous Insufficiency. Phlebology. 2020;35(5):337-44.
- Parsi K, Roberts S, Kang M, et al. Cyanoacrylate Closure for Peripheral Veins: Consensus Document of the Australasian College of Phlebology. Phlebology . 2020;35(3):153-75.
- Langridge BJ, Onida S, Weir J, et al. Cyanoacrylate Glue Embolisation for Varicose Veins - A Novel Complication. Phlebology. 2020;268355520901662.
- Quirce S, Baeza ML, Tornero P, et al. Occupational Asthma Caused by Exposure to Cyanoacrylate. Allergy. 2001;56(5):446-449.
- Shaĭdakov EV, Mel'tsova AZ, Porembskaia OI, et al. Experience with using cyanoacrylate glue in endovascular treatment of varicose veins. Angiology and vascular surgery. 2017;23(4):62-7.
- Sato M, Inomata N, Aihara M. A case of contact dermatitis syndrome caused by Dermabond®, followed by contact dermatitis caused by false eyelash glue and Aron Alpha® glue: possibility of cross-reactions among cyanoacrylates. Contact Dermatitis. 2017;77(6):414-15.
- Bitterman A, Sandhu K. Allergic contact dermatitis to 2-octyl cyanoacrylate after surgical repair: Humidity as a potential factor. JAAD Case Rep. 2017;3(6):480-81.
- Sinha KR, Duckwiler G, Rootman DB. Urticarial reaction following endovascular embolization of an orbital arteriovenous malformation (AVM) with n-butyl cyanoacrylate (nBCA) glue. IntervNeuroradiol. 2017;23(6):666-68.
- Haslinda AR, Azhany Y, Noor-Khairul R, et al. Cyanoacrylate tissue glue for wound repair in early posttrabeculectomy conjunctival bleb leak: a case series. Int Med Case Rep J. 2015;8:145-50.
- McDonald BS, Buckley DA. Severe dermatitis from Dermabond® surgical glue. Br J Dermatol. 2014;170(3):739-41.
- Lindström I, Suojalehto H, Henriks-Eckerman ML, et al. Occupational asthma and rhinitis caused by cyanoacrylate-based eyelash extension glues. Occup Med (Lond). 2013;63(4):294-97.
- Shanmugam S, Wilkinson M. Allergic contact dermatitis caused by a cyanoacrylate-containing false eyelash glue. Contact Dermatitis. 2012;67(5):309-10.
- Bruze M, Björkner B, Lepoittevin JP. Occupational allergic contact dermatitis from ethyl cyanoacrylate. Contact Dermatitis. 1995;32(3):156-59.
- Thomsen GF. [Occupational Asthma Induced by Cyanoacrylate Glue]. UgeskrLaeger. 1994;156(36):5131-32.
- Carlson AN, Wilhelmus KR. Giant papillary conjunctivitis associated with cyanoacrylate glue. Am J Ophthalmol. 1987;104(4):437-38.
- Kopp SK, McKay RT, Moller DR, et al. Asthma and rhinitis due to ethylcyanoacrylate instant glue. Ann Intern Med. 1985;102(5):613-15.
- Watts TJ, Thursfield D, Haque R. Allergic contact dermatitis caused by VenaSeal tissue adhesive. Contact Dermatitis. 2019;80(6):393-95.
- Zierau. Ist der Venenklebergefährlich? 2016.
- Zierau UT. Special case: sealing veins with HIV – Der besondere Fall: Venenklebenbei HIV. 2017.
- Thum J. Single Center Erfahrungenaus 234 Stammvenenbehandlungenmitviskösem n – Butyl – 2 – Cyanoacrylat- Verwendbarkeit, Akzeptanz, Ergebnisse; vasomed 1. 2019; 28–31.
- Neff P. ErfahrungenmitviskösemCyanoacrylat in der Niederlassung – Patientenauswahl und Ergebnisse; vasomed 1. 2019; 31–3.
- Operationsverfahren.2019.
- MitinnovativerTechnikgegenVenenleiden. 2018.
- Zierau UT. SaphenionScience – AllergieVenenkleber? Update. 2020.